Why the greenhouse effect does not violate the first law of thermodynamics
| 14-09-2016 23:51 | |
| jwoodward48★★★★☆ (1537) |
Into the Night wrote:jwoodward48 wrote: It is fitting that you call yourself the Parrot Killer, since once all parrots are dead, you only have thinkers to talk with. CO2 doesn't really obstruct anything. While it can absorb certain narrow bands of IR light, the effect of that absorption is the destruction of the photon and conversion of the energy it contained into kinetic energy (temperature). This is true for a single molecule or a whole beaker full of the gas. This is an obstruction of the escaping light, no? The atmospheric density of CO2 is currently around 0.04% of the atmosphere. That means the energy the CO2 gained by absorbing a photon is dissipated across approx 2500 other nearby molecules of other stuff. This dissipation happens by conduction. That in turn is conducted and convected away from the warmer surface below. It doesn't matter where the energy goes as long as it's not escaping. If you were able to see infrared directly, you see the surface solid and 'hot'. The air above it in a 'redder' dim glow like a fog, that becomes redder and dimmer as you rise in to the sky. Quite true. I agree with all of this. The reason the light dims is because there are fewer molecules as you rise into the sky. The reason you 'see' the light at all is because each molecule is putting out photons according to it's temperature, just as Stefan-Boltzmann predicts. Ditto. Except just a pedantic criticism - didn't you say that I could see infrared? The reason such a dim glow cannot warm the surface is because of the 2nd LoT, which states that heat always flows from hot to cold, at a rate determined by the difference. It never flows from cold to hot. When a dam is constructed, how does the dam at the shallow part make the other part deep? The 2nd law can't be applied as easily when you have constant incoming and outgoing energy, like a heat-leaky house with a shivering owner. Is there some crucial difference between CO2 and a dam? Is there some reason that an uninsulated home cannot represent the Earth? It takes energy to emit a photon from the Earth. When that photon strikes a gas and is absorbed, all of that energy is converted to kinetic form. As far as the electromagnetic energy is concerned here, it is lost completely. But it warms it more than "no photon"! There is a net loss of energy to space, yes, but what matters is incoming energy minus outgoing energy. No matter how slightly the second is decreased, it will still have an effect on the equilibrium. The 2nd LoT can be extended to all forms of energy. High energy areas 'flow' to low energy areas. The reverse cannot happen. This law, which is the law of entropy, affects all forms of energy, not just kinetic (thermal) energy. But the energy is flowing to space. It's just that more energy is coming from the Sun, and not quite enough energy has left to make "enough room" in the current temperature. It's like a hotel - if it's scheduled to be completely full on Monday and Tuesday, and one guest who was supposed to leave on Monday decided to stay, the hotel would be too full. There is indeed a net outflow of energy when looking solely at the outgoing energy, but incoming energy matters just as much. Energy cannot be created or destroyed. So the effect is that a concentrated bit of energy will be dissipated over a larger and larger area. The same energy is there, but spread over such a large area as to be effectively useless. @2ndLoT: But that's just it! It takes energy to reduce entropy in a location, and that energy comes from the Sun. Besides, the 2ndLoT is dark magicks and I never trusted it anyway. [/jk] |
| 14-09-2016 23:53 | |
| Into the Night (21599) |
jwoodward48 wrote: No. It does, however, ignore the effect of thermal energy passing right through. Remember, of all the energy coming from the Sun, the bulk of it is IR. Only the peak energy is in the visible region. The reason is that IR is such a wide frequency band. It's like comparing the AM broadcast band (500kHz to 1600kHz) against the entire shortwave band (300kHz to 30000kHz) (what the FCC calls the High Frequency band). The land, the sea, the air, are primarily heated by IR. Visible light does very little heating. It's energy is too great. Much of it is just reflected off. In other cases, absorption does not cause heating, it causes bonds to break and ionization. As frequency increases into ultraviolet, the energy is so great as to cause some serious ionization and molecular bonding damage. Visible light is why all plastics break down when exposed to daylight (or even roomlight for long enough!) over time. Rubber hardens. Photographic film emulsion crystallizes. Electric eyes in elevator doors can 'see' you walk through the door. If you put up a barrier to IR, you would block incoming radiation that is capable of easy conversion to thermal energy as well as outgoing thermal energy. Even if visible light came through, it would not warm the surface anymore than a sky full of LEDs. (Now THERE's a concept!) Consider the lowly LED. It puts out visible light (a few even put out ultraviolet light!) that you can see. It is not as hot as an equivalent incandescent light bulb. You don't feel heat from it. It is just the light and nothing else. If you put a white LED in a closed container and measure the temperature increase, then do the same with an incandescent bulb of the same brightness, you will find the LED to much cooler. The reason is because the incandescent bulb produces light the black body way. The Stefan-Boltzmann way. It produces visible light by using a high temperature. The LED, on the other hand is like playing the violin, not burning it. It can emit light by raising electron orbital energy and letting it drop again. Each time an electron drops, a photon is emitted. Electrons shifting between orbital energies is not temperature. It is not kinetic energy in the classical sense at all. It is a quantum leap. This is the so-called harmonic absorption and emission of energy and it has nothing to do with temperature at all. It does not change the temperature of the molecule, and it is not emitted by the temperature of a molecule. A molecule, when hit like this, will simply spit the photon back out in pretty short order. The photon goes on its merry way with affecting the molecule itself. That molecule is still the same temperature it was before. Nothing was converted into thermal energy, and the effect is like deflecting the photon. Energy is still dissipated, however. The 2nd LoT still applies. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 14-09-2016 23:55 | |
| Into the Night (21599) |
jwoodward48 wrote: Actually, this forum has pretty decent editing ability. There are quite a few that give you none at all. You can probably imagine how those who like to pick apart misspellings or funky wording have a field day on such a forum. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 14-09-2016 23:56 | |
| jwoodward48★★★★☆ (1537) |
Into the Night wrote:jwoodward48 wrote: I am talking about heat. Incoming light gives heat, outgoing light takes away heat. (Or thermal energy, whichever.) A good way of thinking about this is a bathtub. If I leave the drain open and the faucet on, it will find equilibrium, will it not? There will be a point when the height of the water produces an outflow that is equal to the inflow. Individual water comes and goes, but the overall level is constant. But then suppose that I shove the drain-plug in at an odd angle. Now the drain can only work at 50% efficiency. Now the outflow is 50% of the inflow, and equilibrium is lost again. The water level increases, whether slowly or rapidly, and eventually the pressure at the bottom is twice what it used to be. Now the outflow equals inflow again, and equilibrium returns. |
| 15-09-2016 00:06 | |
| Into the Night (21599) |
jwoodward48 wrote: They are both related. Such graphs often have a 'temperature' scale associated with the color. In fact that is how photographers tend to refer to color and light. That scale is generated out of the Stefan-Boltzmann law. Some of these graphs also have a scale showing relative energy (more rare). Those scales are generated using Planck's law. Most of these graphs have neither scale. They only relate a frequency to color, or the wavelength (the inversion of frequency) to color. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 00:08 | |
| Into the Night (21599) |
jwoodward48 wrote:Into the Night wrote:jwoodward48 wrote: Again, this is ignoring the effects of thermal energy, which aren't affected by such mirrors. S-B applies to the atmosphere as well as the surface. It applies to all matter no matter where it's location. It even applies to a lonely whiff of hydrogen you come across in open space. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 00:14 | |
| jwoodward48★★★★☆ (1537) |
...Are you talking about thermal radiation? Because electromagnetic radiation should be affected by this mirror. Just in case, though, let's make it "magic" for now. First I need to make sure that it's actually physically possible for anything to heat up the Earth. Okay. S-B does apply everywhere. But if part of the radiation is blocked... so what? How exactly are you invoking S-B? |
| 15-09-2016 00:50 | |
| Into the Night (21599) |
jwoodward48 wrote:Into the Night wrote:jwoodward48 wrote: I consider anyone that dissolves into insult streams with no arguments presented to be a dead parrot. I do not consider fallacies to be arguments. You are far from that! You are not just parroting, and you are presenting clear and defined arguments and questions. While occasional fallacies do occur (no one is completely free of them!), they have been very minor and not worth bringing up. Yes, it is certainly my hope to have only thinkers to talk to. Call it my ideal that I know will never be. Again, I thank you for keeping the conversation on this level. jwoodward48 wrote:CO2 doesn't really obstruct anything. While it can absorb certain narrow bands of IR light, the effect of that absorption is the destruction of the photon and conversion of the energy it contained into kinetic energy (temperature). This is true for a single molecule or a whole beaker full of the gas. It IS the obstruction of light (from whatever direction it comes from), but not the obstruction of energy. Conversion of electromagnetic energy to kinetic energy is energy 'lost' as far as electromagnetic energy is concerned. All of the absorbed energy is converted to another form that easily makes it past the 'obstruction'. It is the 'obstruction' that allows this to happen. jwoodward48 wrote:The atmospheric density of CO2 is currently around 0.04% of the atmosphere. That means the energy the CO2 gained by absorbing a photon is dissipated across approx 2500 other nearby molecules of other stuff. This dissipation happens by conduction. That in turn is conducted and convected away from the warmer surface below. My point is simply that it is escaping. If you put up a barrier of any kind, the stuff slips around you like a ghost. It's a 'shape shifter'. It can convert to another form of energy and back again. jwoodward48 wrote:If you were able to see infrared directly, you see the surface solid and 'hot'. The air above it in a 'redder' dim glow like a fog, that becomes redder and dimmer as you rise in to the sky. Yes I did. This is what you would see. jwoodward48 wrote:The reason such a dim glow cannot warm the surface is because of the 2nd LoT, which states that heat always flows from hot to cold, at a rate determined by the difference. It never flows from cold to hot. Yes. It is because even an uninsulated house is insulated...by its walls and roof. Any enclosed container (even an uninsulated house) limits convective heat loss (or gain). The difference between CO2 and a dam is that CO2 simply converts energy to something else, while the dam does not. The dam is only concerned with river flow. Energy is not like a river. The house does not really apply because a house has some kind of furnace inside it and CO2 does not act like an insulator. jwoodward48 wrote:It takes energy to emit a photon from the Earth. When that photon strikes a gas and is absorbed, all of that energy is converted to kinetic form. As far as the electromagnetic energy is concerned here, it is lost completely. Except that you are talking about light, not temperature. There is no equilibrium with light. Light moves too fast for that, and light is not heat. jwoodward48 wrote:The 2nd LoT can be extended to all forms of energy. High energy areas 'flow' to low energy areas. The reverse cannot happen. This law, which is the law of entropy, affects all forms of energy, not just kinetic (thermal) energy. Whether you 'trust' it not doesn't change the law, it only changes your acceptance of it. All scientific theories are falsifiable. The 2nd LoT is no exception. Even though we call it a law, it is still just a theory and will remain so until it is destroyed by another theory. Remember, to gather the dissipated energy, you must spend an equal amount of energy doing it. The Sun is not gathering dissipated energy. It is dissipating it, just as the Earth does. Once it's gone, it's gone. It too is subject to the 2nd LoT. Using energy from the sun, we can indeed 'reduce' entropy in localized ways. We can use it to manufacture fuel that can later be burned. We can use it to vaporize the sea, let it fall as rain, and capture the runoff using dams. We can use it to trigger chemical reactions, such as what happens in a plant. The total energy, however, remains the same flow. We cannot recover the dissipated energy overall. It would take an equal amount of energy to do it, and we would be right back where we started. Not even the Sun has that much power. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 00:53 | |
| Into the Night (21599) |
jwoodward48 wrote:Into the Night wrote:jwoodward48 wrote: It might seem that way, but you are not. Heat is strictly defined as the flow of thermal energy. It has nothing to with light at all. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 01:03 | |
| jwoodward48★★★★☆ (1537) |
Hmm. Okay. I meant thermal energy then. I guess it's another "scientist vs layman" use of the word, and after years without a refresher on this, I slipped back to the layman's use. The light itself is only a way of conveying the energy from the Sun to the Earth. The Earth then stores that energy as thermal energy and releases it through black body radiation. Right? If some of the outgoing radiation is instead redirected back toward Earth, then the equilibrium of incoming and outgoing light gets a metaphorical hard kick. Once it restabilizes, the temperature is higher. The bathtub and house analogies are not perfect, but they contain the idea of "inflow/outflow equilibrium" that I'm trying to convey. (also, I do think the 2nd LoT is fine, it's actually the "entropy = chaos" interpretation of it that I dislike. Chaos and disorder are crucial ideas that humanity has, but their layman interpretation cannot quite be quantized like that. In fact, maximum entropy would mean absolutely flat energy, everywhere. Seems "orderly" to me, in the layman's sense of disorder.) Edited on 15-09-2016 01:16 |
| 15-09-2016 01:10 | |
| Into the Night (21599) |
jwoodward48 wrote: No, I am talking about heat flow, which has nothing to do with light. Assuming your magick barrier blocks all electromagnetic radiation coming into the Earth (the only way to keep it from heating the Earth). Such a barrier would also block any electromagnetic radiation from leaving the Earth, and any ability for radiated energy to leave the Earth. A perfect insulator from your magick barrier. The result is that the Earth, which contains a molten core (the result of fission inside the Earth), would gradually get hotter and hotter. The whole Earth would eventually look a lot like it's present mantle. This is the same as putting such a perfect insulator around a house and turning the furnace on. You will eventually burn down the house (and the furnace) that way. Once the fuel is consumed and the fire is out, the remains will glow with the heat of it forever. (why do I suddenly feel like Randall Munroe describing a fastball pitched at near the speed of light?) The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 01:44 | |
| IBdaMann (14414) |
jwoodward48 wrote:The Earth then stores that energy as thermal energy and releases it through black body radiation. Right? Nope. No substance stores thermal energy. No substance traps thermal energy. Thermal energy pours (radiates) freely out of all substances based on its temperature (re:Stefan-Boltzmann) jwoodward48 wrote:If some of the outgoing radiation is instead redirected back toward Earth, then the equilibrium of incoming and outgoing light gets a metaphorical hard kick. Nope. Energy can change form but there is no magical direction that somehow creates more of it (re: 1st LoT) jwoodward48 wrote:Once it restabilizes, the temperature is higher. Wow! You are still kicking Stefan-Boltzmann to the curb, I see. P . U . R . E . * . S . C . I . E . N . C . E . * . D . E . N . I . A . L Wake me up when you get to the point where you resort to insisting that the constants of emissivity and albedo are actually variables whose value "greenhouse gas" magically modifies to the value you wish it were. You're not there yet but it should be just around the corner. . I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 15-09-2016 02:16 | |
| jwoodward48★★★★☆ (1537) |
Into the Night wrote:jwoodward48 wrote: ...isn't radiative heat in the form of electromagnetic radiation? Hot glowing iron - the light carries some thermal energy away, converting it to some other type of energy (electromagnetic, I assume?). Assuming your magick barrier blocks all electromagnetic radiation coming into the Earth (the only way to keep it from heating the Earth). But that's the point! It lets any light in, no matter what, but doesn't let any light escape. I'm currently ignoring physical properties and such in order to figure out if, given a "perfect material," I could heat up the Earth. Such a barrier would also block any electromagnetic radiation from leaving the Earth, and any ability for radiated energy to leave the Earth. A perfect insulator from your magick barrier. Oops, I think I may have stressed the post wrong. Only half of the energy is prevented from leaving. It would prevent any and all radiation from leaving North America, but not India, for instance. The result is that the Earth, which contains a molten core (the result of fission inside the Earth), would gradually get hotter and hotter. The whole Earth would eventually look a lot like it's present mantle. See previous posts. Some energy cannot escape, not all. This is the same as putting such a perfect insulator around a house and turning the furnace on. You will eventually burn down the house (and the furnace) that way. Once the fuel is consumed and the fire is out, the remains will glow with the heat of it forever. You like xkcd too? Awesome! Yeah, this does feel like an explainxkcd - an implausible situation being used to convey scientific explanations and theories. |
| 15-09-2016 02:18 | |
| Into the Night (21599) |
jwoodward48 wrote: Again, this is assuming that all photons are equal. They are not. This is an important concept which is why IBDaMann keeps harping on it. It can't be ignored. It blows away any common accounting you try to do with equal photons. jwoodward48 wrote: In a way, it all comes down to what you define 'order' or 'chaos' as. Say you have a transparent box full of red bugs, and a transparent box full of black bugs. Entropy is at a minimum. You have two boxes each with their own unique color of bugs. Connect a tube between them, and let the bugs crawl back and forth between the boxes. Eventually you have two boxes, each filled with the same amount of bugs, but now each box contains generally an equal amount of red and black bugs. Entropy is at its maximum. Entropy is just a measure on how difficult it would be to restore the original order of the boxes before you connected the tube. So it can be said that entropy is a definition of order vs the uniformity of chaos. The same thing happens with energy. Once you 'let it out of the box', so to speak, it takes a lot of work to get it back into the 'box' it came from. In fact it takes an equal amount of energy to do so. This is the reversibility of the Carnot engine. Yeah, you can power the engine by using heat, but you can also put the heat back by powering the engine. If course it takes an outside source of energy to do that. Our modern refrigerators and air conditioners do just exactly that. They are built like engines, but we put power into them rather than extracting power out of them. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 02:31 | |
| Into the Night (21599) |
jwoodward48 wrote:Into the Night wrote:jwoodward48 wrote: Light is light and thermal energy is thermal energy. Never the twain shall meet and be the same thing. You can convert one to the other, but they are not the same thing. jwoodward48 wrote:Assuming your magick barrier blocks all electromagnetic radiation coming into the Earth (the only way to keep it from heating the Earth). It truly would be a magick substance to have the properties you need. There is no such substance, not even remotely like it. Carbon dioxide certainly has none of these qualities, neither does water vapor. jwoodward48 wrote:Such a barrier would also block any electromagnetic radiation from leaving the Earth, and any ability for radiated energy to leave the Earth. A perfect insulator from your magick barrier. You are still ignoring the effects of thermal energy (which is not light at all). You are also still ignoring the different energies of different photons. jwoodward48 wrote:The result is that the Earth, which contains a molten core (the result of fission inside the Earth), would gradually get hotter and hotter. The whole Earth would eventually look a lot like it's present mantle. If you open the door to energy escaping, the hotter the surface, the faster the energy will escape until the energy inside the barrier is the same as the energy outside the barrier. Since no sun is allowed in this scenario, other than what comes through the same door, the incoming energy to replace what is lost to space is minimal. The Earth will be quite cold, but warmer than space. jwoodward48 wrote:This is the same as putting such a perfect insulator around a house and turning the furnace on. You will eventually burn down the house (and the furnace) that way. Once the fuel is consumed and the fire is out, the remains will glow with the heat of it forever. I'm not as good an artist. My experiences in this stuff come from a different direction as well. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 03:16 | |
| jwoodward48★★★★☆ (1537) |
IBdaMann wrote:jwoodward48 wrote:The Earth then stores that energy as thermal energy and releases it through black body radiation. Right? You are the funniest. Thermal energy literally cannot be anywhere but stored within a substance. You are literally the most wrong it is possible to be. So air does not hold thermal energy? How come I don't burn by day and freeze by night then? Thermal energy pours (radiates) freely out of all substances based on its temperature (re:Stefan-Boltzmann) Indeed! But that takes time, and in the meantime the heat exists within the substance. When I boil water and then take it off the stovetop, it doesn't immediately stop boiling. Matter holds heat. Tell me, have you ever taken Intro to Chem? These are basic concepts. jwoodward48 wrote:If some of the outgoing radiation is instead redirected back toward Earth, then the equilibrium of incoming and outgoing light gets a metaphorical hard kick. Are you, good sir, hard of seeing? Hast thee sight? Are you bloody blind? I'll assume that an enormous fly landed on your screen and prevented you from reading the explanation I literally posted a few hours ago, and rewrite it. === Energy comes from the Sun. [citation needed] This energy is in the form of electromagnetic radiation, in accordance with the Black Body Laws (BBL). The light approaches the Earth, and, let's say, 5% of it (not actual number) is absorbed. Another 50% of that is radiated away from the Earth, never to be seen again. Thus, the incoming light is 97.5% of what an nonatmospheric Earth would receive. (A nonatmospheric Earth would receive and give off equal amounts of energy, as does an atmospheric Earth in equilibrium. But CO2, as will be seen, disrupts that equilibrium.) Now the light hits the Earth, heating it up by exciting its atoms. The Earth then radiates this energy away over time, not immediately, as per the BBL. Some of the energy that is radiated away is intercepted. Since the Earth is cooler than the Sun [citation needed], the Earth's black body radiation has a longer wavelength. This wavelength is more susceptible to being absorbed by greenhouse gases, as seen in this graph: 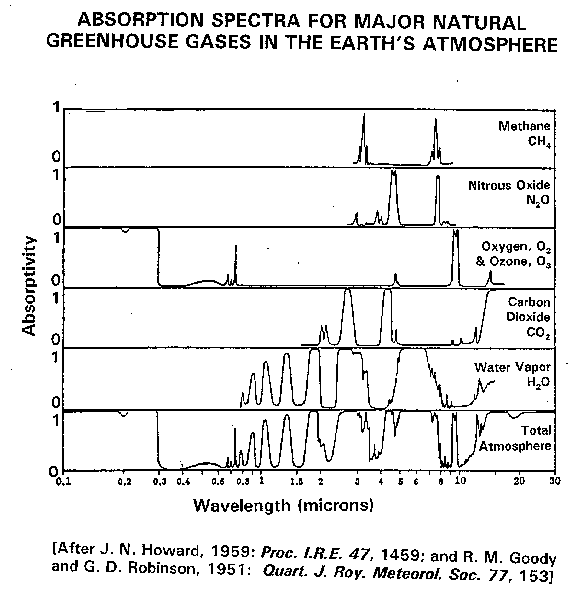 And the black body curves are here (yellow is Sun):  This means that instead of 5% being intercepted, maybe 15% is. Another 50% of that 15% is still radiated out, but the 7.5% stays in the system (for now). The out-flowing energy is thus 92.5% of what a nonatmospheric Earth would give off. Since the energy flowing out is less than the energy flowing in, we have a problem! The energy has to stay in the Earth (the 1st LoT says it can't just disappear), so the temperature increases. All is not lost, though. A runaway reaction must still inevitably halt, for as temperature increases, so does radiation! (As per Planck's Law.) Once the out-flowing energy equals the in-flowing energy, equilibrium will be reached. By that point, though, who knows how warm the Earth will be? (Scientists do. I don't have the numbers, so I can't estimate that.) But the situation is still dire. As the temperature increases, ice caps will melt, decreasing Earth's albedo and releasing additional CO2. The first will produce a greater incoming energy rate, and the latter will decrease the outgoing energy rate more than it will decrease the incoming energy rate. This exacerbates the warming - a feedback loop. Note that there is never a net flow of energy from cold to warm - just a decrease in the decay-motion of energy from warmth to coldness. This does not violate the second LoT. Also note that this incorporates the 1st LoT. The energy required to heat the Earth comes from the Sun. More specifically, energy is constantly flowing in and out of the Earth. If energy-bits spend more time within the Earth, a greater temperature is the result. The incoming energy is always the same - heck, it sometimes decreases! Some outgoing energy is just redirected toward heating the Earth. jwoodward48 wrote:Once it restabilizes, the temperature is higher. Wow! You are still not understanding that radiation can be absorbed and redirected back to Earth, still following S-B! S-B just says that X amount of energy will leave Earth's surface - it does not care if some of that energy returns.[/quote] Wake me up when you get to the point where you resort to insisting that the constants of emissivity and albedo are actually variables whose value "greenhouse gas" magically modifies to the value you wish it were. Wake me up when you're posting for the purpose of rational debate, not a chance to insult people and say "I'm so much better than you." You're not there yet, and I doubt you'll ever reach it. You never accept anyone's arguments, but you don't even contribute to a conversation, spend one second being polite, or explain how everyone else is so obviously, painfully wrong. But once you get past this, give the exact parts of my argument that violate each specific law you claim that I have violated. Attached image:  |
| 15-09-2016 03:33 | |
| jwoodward48★★★★☆ (1537) |
Into the Night wrote:jwoodward48 wrote:Into the Night wrote:jwoodward48 wrote: You were saying that thermal energy cannot be blocked as light is. But the only effective way for energy to escape from Earth is by radiation, which is light. jwoodward48 wrote:Assuming your magick barrier blocks all electromagnetic radiation coming into the Earth (the only way to keep it from heating the Earth). I agree. This is a very idealised substance, but if I can get the slightest imbalance between incoming-energy-absorption and outgoing-energy-absorption, a similar but weaker effect is created. I am fairly sure that carbon dioxide blocks outgoing energy better than incoming energy. jwoodward48 wrote:Such a barrier would also block any electromagnetic radiation from leaving the Earth, and any ability for radiated energy to leave the Earth. A perfect insulator from your magick barrier. Thermal energy is not going to space. Thermal energy is the "storage form" of energy until it can be radiated to space again in the form of light. Could you explain how the different energies of specific photons affects the macroscopic, overall energy flow? I can't really see how it makes a difference. I'm looking at the total energy flows - how does an individual photon's energy play into all this? (If it does matter, it sounds incredibly interesting - I'd like to know how that works.) jwoodward48 wrote:The result is that the Earth, which contains a molten core (the result of fission inside the Earth), would gradually get hotter and hotter. The whole Earth would eventually look a lot like it's present mantle. Ahh, but the one-way-ness of the door matters. Without the one-way-ness, those problems you noted would definitely happen. jwoodward48 wrote:This is the same as putting such a perfect insulator around a house and turning the furnace on. You will eventually burn down the house (and the furnace) that way. Once the fuel is consumed and the fire is out, the remains will glow with the heat of it forever. Hard to beat his stick figures. |
| 15-09-2016 08:45 | |
| Into the Night (21599) |
jwoodward48 wrote:Into the Night wrote:jwoodward48 wrote:Into the Night wrote:jwoodward48 wrote: Not quite. There are three problems with this. One relates to thermal energy, the other relates to Planck's law and Stefan-Boltzmann's law. One way to measure air density is by it's pressure. At sea level, the air pressure is normally around 1000 millibars (about 29.92 inches of mercury). At this point, all of the atmosphere is above you, pressing down on you with a pressure equivalent to 14.7 psi. (all of this is assuming a 'standard' day at a 'standard' temperature as determined by the FAA and U.S. Meteorological Service.) Rising into the sky, pressure drops. By the time you reach about 15000 ft (or flight level 150 or about 3 miles), pressure has dropped to only 500mB, or only half that found on the surface. This means that half of the mass of the atmosphere is below you as there is above you. The troposphere, which has convective and conductive activity, both ways of moving heat thermally only, ends at about 30000 to 40000 ft (depending on your latitude.). At this altitude, pressure drops to approx 300mB, or only 1/3 what it was at the surface. There is now 2/3 of the atmosphere below you as above you (by mass). Here convective heat stops, leaving conduction and radiation as the sole means to lose energy. Conduction for thermal energy, and radiation for any thermal energy that is reconverted into light. Here the atmosphere is about the coldest you will see for a long time as we rise into the sky. Any radiation will be correspondingly weak, due to the cold air (typically -60 deg F). We are also past 2/3 of any blocking carbon dioxide (actually slightly more, since carbon dioxide has a tendency to sit lower in the atmosphere due to the weight of that molecule). Conduction and convection can easily carry thermal energy this far alone. A tremendous amount. From here going up, temperature begins to rise again. As it rises, the pressure drops still further. At the top of the stratosphere, you reach the stratopause. This rising temperature is due to the formation and destruction of ozone from oxygen. The formation takes place best near the bottom of the stratosphere (an energy sucking reaction as ozone is a higher energy molecule than oxygen is), and is destroyed again near the top (by natural decay and harsher UV light) releasing heat as part of that reaction. We are now 32 miles high. Pressure has dropped to only 1mB, but the temperature has risen to 40 deg F. At this height, there just aren't as many molecules. Total energy is the sum of molecular activity in a given volume, so total energy has dropped even though temperature has increased. Conduction between molecules sucks right now, and conversion to radiation is predominant. We are of course, now above 99% of the atmosphere and any blocking CO2. Now the temperature begins to fall again as we rise into the sky still further. Through the mesosphere and the thermosphere, radiation is the predominant way of losing energy. The second problem is that carbon dioxide may absorb the light (even at low altitude and thus blocking it), but that absorption occurs only once. The excited molecule will not absorb light again until it loses that energy in some way. Fortunately, there is an easy way to shed that energy. Conduction of the resulting thermal energy into surrounding air. Once done, it can absorb another photon, and its lather, rinse, repeat. This means that while it may block IR light, it just converts it to thermal energy (you can't destroy energy) which rises along with the rest of any hot air. The third problem is that any photon emitted from carbon dioxide will be due to the application of the Stefan-Boltzmann law. This will be a lower energy photon than the one that was absorbed due to Planck's law. Even if it strikes the surface and is reabsorbed, the surface will not reach the temperature it lost when it emitted the original photon. This is where we come to an important point. Photons are not equal. A given photon does not carry the same energy as every other photon. This difference is central to understanding the 2nd LoT in terms of electromagnetic energy as an isolated system. Between the difference of electromagnetic energy, the loss of atmosphere as we rise in to the sky (and the resulting bulk of CO2 we bypass by using thermal energy transfer (heat), and conversion of light absorbed by CO2 into thermal energy (only helping to bypass it, so to speak), Earth can cool quite efficiently without worrying about 'blocking' CO2 (or any other 'greenhouse' gas). jwoodward48 wrote:jwoodward48 wrote:Assuming your magick barrier blocks all electromagnetic radiation coming into the Earth (the only way to keep it from heating the Earth). Rather than having me teach that to you, May I recommend you research Planck's law, including how it was created (Hint: Start with Einstein's investigation into the photoelectric effect.) Looking at how Max Planck arrived at his equation is not only a fascinating way of following the reasoning that brought us here, but often into the personalities of the scientists themselves as people. I have shown you how the law applies to the atmosphere in my response above. Now is the time for you to research this question. jwoodward48 wrote:jwoodward48 wrote:The result is that the Earth, which contains a molten core (the result of fission inside the Earth), would gradually get hotter and hotter. The whole Earth would eventually look a lot like it's present mantle. Quite true. There is no substance, however, that acts in only one direction. Not even CO2. As I said, the bulk of the surface is warmed by infrared energy from Sun, not visible light. There really is no way to separate the direction of light by carbon dioxide. The stuff simply reacts to IR the same way the land and the sea does. jwoodward48 wrote:jwoodward48 wrote:This is the same as putting such a perfect insulator around a house and turning the furnace on. You will eventually burn down the house (and the furnace) that way. Once the fuel is consumed and the fire is out, the remains will glow with the heat of it forever. I think he is best with his comics about some aspect of Unix (or Linux). He has a few classics outside of that, like comic 1052. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 15-09-2016 14:50 | |
| IBdaMann (14414) |
jwoodward48 wrote: Could you explain how the different energies of specific photons affects the macroscopic, overall energy flow? So NOW you want to steer back to Stefan-Boltzmann? If you want to discuss photons then work with Planck's Law. If you want classic physics, work with Stefan-Boltzmann. Stefan-Boltzmann can be derived from Planck's distribution. . I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 15-09-2016 15:27 | |
| jwoodward48★★★★☆ (1537) |
But doesn't SB apply everywhere? Re: "here's where to look for the answers": thanks, that's even more fun! I couldn't really explain my problem to Google effectively, or I'd have done that first. Edited on 15-09-2016 15:39 |
| 15-09-2016 15:53 | |
| IBdaMann (14414) |
jwoodward48 wrote: But doesn't SB apply everywhere? Yes. So apply it everywhere. . I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 16-09-2016 07:39 | |
| jwoodward48★★★★☆ (1537) |
It doesn't apply to the price of sorium on Luna. Actual meaningful post: I had misinterpreted what you said. My response makes no sense with my current understanding of your post. Just ignore it. |
| 17-09-2016 02:40 | |
| jwoodward48★★★★☆ (1537) |
Wait a minute. Into, you said that the majority of the warmth on Earth comes from infrared. But perhaps even more infrared light would be measured in sunlight outside the atmosphere. Maybe the atmosphere absorbed much of the largest component of the light, but did not diminish its proportion below 50%. |
| 17-09-2016 17:13 | |
| IBdaMann (14414) |
jwoodward48 wrote: I'm curious as to why you fixate on the differing wavelengths, i.e the form of the energy, when the 1st LoT tells you it doesn't matter. Of course with that in mind, Stefan-Boltzmann tthen tells you the temperature therefore won't change. Jussayn. . I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 17-09-2016 18:57 | |
| jwoodward48★★★★☆ (1537) |
FFS, the first LoT is not violated! You want to convince me of that, show me your equations and reasons why you think that. |
| 17-09-2016 20:38 | |
| IBdaMann (14414) |
jwoodward48 wrote:FFS, the first LoT is not violated! FFS, you apparently MUST deny science AND pout like a ****ing baby. So what now? Is it I that is insulting you? I'll assume so and just get my money's worth: FFS. go learn thermodynamics. You're a ****ing ignorant moron and you pout on top of it. Hey idiot, anytime you claim that energy changing form is resulting in more energy then FFS you are violating the 1st LoT. If you are saying that temperature is increasing then FFS you are saying there is more energy. FFS, learn basic logic, basic science, basic math and all the other things required for normal cognitive function. FFS, stop denying science just because someone indoctrinated you into religious slavery. jwoodward48 wrote: You want to convince me of that, FFS, only you can convince you, ... and that simply won't happen as long as you are having someone else do your thinking for you. Let's run through your bullshit: we avoid Stefan-Boltzmann and instead obsess on particular wavelengths and say that if they go in the right direction then there becomes more energy (increase of temperature). You're a moron. . I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 17-09-2016 20:46 | |
| IBdaMann (14414) |
jwoodward48 wrote:. Into, you said that the majority of the warmth ... Wow, you don't need to get so technical. . I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 17-09-2016 22:44 | |
| jwoodward48★★★★☆ (1537) |
You're a moron. Your naive interpretation of the scientific laws is only supported by your vitriolic hatred for everyone who doesn't bow down to your "expertise." You don't have any actual backing argument, just and mis-invocations of various basic laws and managing to misinterpret every argument that comes your way. Here's an explanation that should be able to get through your thick skull: Some of the energy radiated by the Earth is absorbed by the atmosphere and radiated back to Earth, increasing the incoming energy measured at the surface. See? The first law is not violated. We can argue whether the phenomenon is sufficient enough to noticeably heat the Earth, we can argue whether the atmosphere is saturated with CO2, but arguing that global warming violates the 1st law is just ****ing stupid. Here's another explanation: "To summarise: Heat from the sun warms the Earth, as heat from your body keeps you warm. The Earth loses heat to space, and your body loses heat to the environment. Greenhouse gases slow down the rate of heat-loss from the surface of the Earth, like a blanket that slows down the rate at which your body loses heat. The result is the same in both cases, the surface of the Earth, or of your body, gets warmer." Also, note that Into said this: "As I said, the bulk of the surface is warmed by infrared energy from Sun, not visible light." My response was: "Wait a minute. Into, you said that the majority of the warmth on Earth comes from infrared." That is a perfectly fine response for someone typing tersely on their phone. It was not a complete sentence, it was a reference to a complete sentence that was posted a few posts before and that could be referenced if any confusion came up. For someone who hates science, you sure do insult anyone who varies from your idea of "perfect science-talk". |
| 07-01-2018 14:15 | |
| casualreader☆☆☆☆☆ (4) |
old thread, but just 2 cents 1st law of thermodynamics? https://en.wikipedia.org/wiki/First_law_of_thermodynamics energy is conserved - no energy is lost there isn't any mistake about that planck's law ?  at room temperatures, hardly anything radiates, very low radiation energy (the room temperature radiation peak is around that 10.5um on the graph) and space is pretty much vacuum literally an insulator (non-conductor, non-convector) for heat the earth is 2/3 covered by oceans  all that radiation from the sun in the form of infrared radiation is absorbed in a big way by the oceans, radiation in the band 1000nm - 100um infrared radiation from the sun is simply absorbed / swallowed by the oceans that's somewhat less severe without carbon dioxide, a bigger fraction of that solar radiation gets *reflected* back into space, but with carbon dioxide http://clivebest.com/blog/?p=1169  ^^ carbon dioxide doubles down on infra-red radiation absorbed into the earth, it does so by absorbing infrared radiation in the 10nm range, increasing scattering so that even more of that radiation is absorbed, and perhaps it is not simply carbon dioxide but all sorts of industrial emissions that cause fog, smog, all that industrial emissions like steam, dust, and everything else severely increase atmospheric scattering so that much less of that sun's radiation is reflected back into space, and all that energy is absorbed into the earth so you see giant ice shelfs size of entire countries in Antarctica breaking off and melting into the sea https://youtu.be/jDXr98pt0bg https://youtu.be/z5x0qwhZDuM and the Artic nearly free of ice in summer https://www.youtube.com/watch?v=lZYIbRS7jG4 it won't be too hard to guess that the Artic may no longer have ice in summer in a near future and that Antarctica melts the earth's atmosphere gets cooler? yup because Antartica melts that absorbs all the heat into the melting ice cooling down the atmosphere, while the Antartica continues its melting himalaya melts https://youtu.be/hPa_m5qNF6s and in a not too distance future perhaps mount everest may no longer be capped by snow/ice yes that's the most visible effect of greenhouse effect and yes it i real, global warming is real, Antarctica is melting, Arctic has no more ice and Himalayas and all those snow capped mountains will have no more snow Edited on 07-01-2018 15:00 |
| 07-01-2018 15:53 | |
| casualreader☆☆☆☆☆ (4) |
in the last chart above, reposted there is broad spectrum of infrared absorption by *water vapor* then you think about all the steam from cooling towers https://youtu.be/KbxHk7go7UU https://youtu.be/U_z98C188LM you think about all the billions of vehicles on the roads after that gasoline or diesel gets burnt up it simply becomes water vapor and carbon dioxide https://youtu.be/Bj6xNbVEnIQ planes https://youtu.be/ktOOxvFaemU and less visible large ships crossing the oceans that water vapor in the air doubles down on the infrared radiation absorption and all that emissions add further to smog drastically increasing atmospheric scattering, in the end all that infrared radiation gets absorbed by the water vapor, dust etc. all that water vapor forms into clouds turns into rain and add that absorbed solar radiation into the oceans and adding to the oceans' own absorption of solar radiation, hence the oceans warms up https://youtu.be/9-_obMEF_2o https://youtu.be/cbkfqUTRQeI https://youtu.be/aIayxdrX17g and Antarctica, Arctic melts and the snow capped Himalayas (mount everest) become iceless bare rocks Edited on 07-01-2018 15:56 |
| 07-01-2018 16:31 | |
| litesong★★★★★ (2297) |
casualreader wrote:....water vapor in the air doubles down on the infrared radiation absorption....It is good that casualreader shows GHGs do not violate the the 2nd Law of thermodynamics. It is also good to show that water vapor is a phase change, infra-red energy absorbing GHG (whether "natural" or man-made) that is a positive feedback to man-made, non-phase change, infra-red energy absorbing GHGs. |
| 07-01-2018 16:40 | |
| casualreader☆☆☆☆☆ (4) |
a very interesting paper from nasa https://ceres.larc.nasa.gov/press_releases/lectures/Loeb_LaRC_Colloq.pdf |
| 07-01-2018 17:47 | |
| casualreader☆☆☆☆☆ (4) |
litesong wrote:casualreader wrote:....water vapor in the air doubles down on the infrared radiation absorption....It is good that casualreader shows GHGs do not violate the the 2nd Law of thermodynamics. It is also good to show that water vapor is a phase change, infra-red energy absorbing GHG (whether "natural" or man-made) that is a positive feedback to man-made, non-phase change, infra-red energy absorbing GHGs. indeed a very inconvenient truth  and it'd seem there isn't much solution, it is not simply carbon dioxide it is *emissions* and *heat* and plain simple *combustion* from vehicles (gasoline and diesel burns into water vapor and carbon dioxide, etc) and industries that cause the warm water vapor to get into air and the soot in the emissions that cause all that smog + water vapor which cause all the scattering and ultimately absorbing all that infrared and other radiation from the sun, it contributes/leads to a greenhouse effect and global warming Edited on 07-01-2018 17:59 |
| 07-01-2018 22:39 | |
| Into the Night (21599) |
litesong wrote:casualreader wrote:....water vapor in the air doubles down on the infrared radiation absorption....It is good that casualreader shows GHGs do not violate the the 2nd Law of thermodynamics. He didn't. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 07-01-2018 22:42 | |
| Into the Night (21599) |
casualreader wrote: Absorption of light from the surface by CO2 or any other gas or vapor does not warm the Earth. It is just another way for the surface to COOL itself while it heats the atmosphere. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 09-01-2018 15:22 | |
| litesong★★★★★ (2297) |
Absorption of light from the surface by CO2 or any other gas or vapor does not warm the Earth.It warms Earth bio-sphere, & cools other parts of the Earth. Edited by branner on 11-03-2018 11:31 |
Join the debate Why the greenhouse effect does not violate the first law of thermodynamics:
Related content
| Threads | Replies | Last post |
| The "radiative Greenhouse effect" does not exist | 145 | 24-04-2024 02:48 |
| 'Greenhouse' Effect? | 49 | 30-11-2023 06:45 |
| The SCIENCE of the "Greenhouse Effect" | 291 | 05-11-2023 22:46 |
| 17 year old cyclist murdered, do not expect the law to investigate, as the cyclist is always at fault | 0 | 31-07-2023 22:23 |
| Nitrate Reduction - Powerful Greenhouse Gas Emission AND Alkalinity | 102 | 05-06-2023 13:19 |