Greenhouse Gases Do NOT Violate The Stefan-Boltzmann Law
| 28-09-2017 20:44 | |
| James_★★★★★ (2218) |
GasGuzler wrote:James_ wrote: With the warming per decade, there is >> Since 1970, global surface temperature rose at an average rate of about 0.17°C (around 0.3° Fahrenheit) per decade << https://www.climate.gov/news-features/understanding-climate/climate-change-global-temperature This graph is typical, notice from 1910 - 1945 and then 1978 to present, both time frames have a rise of about 0.5° C. I can go into more detail how the Arctic and Antarctica differ. One example is that during an ice age the Arctic will have glaciation that goes as low as the 47th parallel while Antarctica has about the same glaciation as it does now. Antarctica might actually warm during an ice age and then cool when there is no ice age. This suggests that what differentiates between an ice age and a warm period effects the Arctic directly but not Antarctica.; Attached image: 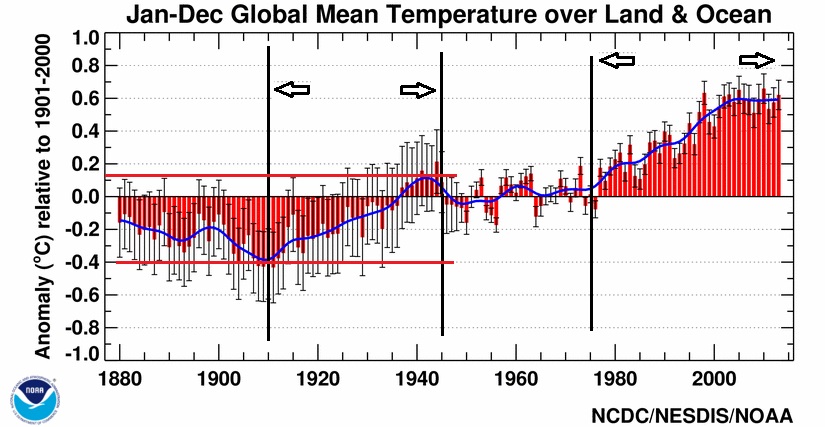
Edited on 28-09-2017 20:47 |
| 28-09-2017 20:55 | |
| James_★★★★★ (2218) |
GasGuzzler, With the Gulf Stream, any hot water from rivers. Poewr plants can raise river temperatures by up to 6° F. In Illinois their river can go over 100° F. during the summer if it's hot enough outside. Slow moving water is also warmer. And it's possible that the glaciers on Iceland and Greenland that melt because of warmer air flowing north can allow for the sea floor to rise as well as allow for more hydrothermal vents. And warm water rising from the ocean floor might slow the Gulf Stream which would cause it to retain more heat. And there are plenty of under water volcanoes as well. https://www.iceagenow.info/underwater-volcanoes-melting-arctic-ice-says-geologist/ I think you'll like this link to NOAA and the IPCC which (about half way down the page) states that; Carbon dioxide (CO2), nitrous oxide (N2O), and methane (CH4) are each important to climate forcing and to the levels of stratospheric ozone (see Chapter 2). In terms of the globally averaged ozone column, additional N2O leads to lower ozone levels, whereas additional CO2 and CH4 lead to higher ozone levels. Ozone depletion to date would have been greater if not for the historical increases in CO2 and CH4. https://www.esrl.noaa.gov/csd/assessments/ozone/2014/summary/ch5.html And why ozone is important, it's about 1 or 2 paragraphs long; Ozone is a gas in the atmosphere that protects everything living on the Earth from harmful ultraviolet (UV) rays from the Sun. http://earth.rice.edu/mtpe/atmo/atmosphere/topics/ozone/o3.html Edited on 28-09-2017 20:56 |
| 28-09-2017 23:19 | |
| Into the Night (21582) |
GasGuzzler wrote:ITN wrote: The only pain you feel is self inflicted. Care to clear any of these? The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 28-09-2017 23:23 | |
| Into the Night (21582) |
James_ wrote: There ARE NO global temperature graphs. There is only manufactured data. James_ wrote: No one has ever recorded the global temperature. James_ wrote: There is no trend without absolute measurements, which have never been done. James_ wrote: It's time to stop getting your BS from a bad movie. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 28-09-2017 23:27 | |
| Into the Night (21582) |
Wake wrote:GreenMan wrote: Argument from randU. It is not possible to determine temperature of the lower atmosphere. Wake wrote:Like yours? Wake wrote:Might as well. Satellites do not have the capability to measure absolute temperature (except for internal components of themselves). Wake wrote: We have no way to measure sea level. There is no valid reference. Wake wrote: Take your own advice, dumbass. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 28-09-2017 23:37 | |
| Into the Night (21582) |
Wake wrote:GasGuzler wrote:James_ wrote: You don't know. It is not possible to determine a global temperature. Science is not links. Wake wrote: Because they believe their manufactured data is better than your manufactured data. Wake wrote: Neither Dr. Spencer nor NASA can determine the temperature of the Earth either. You are just making an argument of randU, the same as the people you are trying to put down. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 28-09-2017 23:38 | |
| Into the Night (21582) |
James_ wrote:GasGuzler wrote:James_ wrote: Argument from randU. It is not possible to determine the temperature of the Earth. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 28-09-2017 23:44 | |
| Wake★★★★★ (4034) |
GreenMan wrote: James, I think you are right about this. And you point out something that I have been trying to get at. I recall Parrot Killer [not too good at it though, because Wake is still alive and doing well] saying something about the emissivity changing, but I wasn't quite sure what he was bitching about, because it wasn't me saying it. He was just twisting something around that I was saying, to make it look like that. But anyway, I don't think the emissivity has to change at all, to accommodate the Global Warming Theory. It stays the same because it is just a number that represents the proportionality between radiation and temperature. It does vary from object to object, due to the amount of radiant energy the object is transforming into thermal energy. Why are you paying any attention to nightmare to begin with? That's simply a losing proposition since it appears that he is a lunatic. You don't let lunatics drag you down. You are correct that emissivity doesn't have to change. All it takes is a tiny change in the sun's emissions which occurs all the time. There is no "proportionality between radiation and temperature". Power in equals power out. That's 1:1 so you don't call it a proportion. Edited on 28-09-2017 23:46 |
| 29-09-2017 00:23 | |
| Into the Night (21582) |
James_ wrote: We first start off with random words strung together in something that almost resembles English. James_ wrote: No, it isn't. Speed of a river does not determine its temperature. James_ wrote: They aren't melting. James_ wrote: You can't slow the Gulf Stream. It is powered by the uneven temperatures of the oceans. If you completely block the northern countercurrents, the Gulf string will still flow. The northern countercurrents will simply move south. James_ wrote: You can't trap heat. James_ wrote: Yes, there is a volcanic ridge under the Arctic sea that was recently active. It seems to be settling down now. James_ wrote: I've stopped searching out propaganda from sites like these. James_ wrote: There is no such thing as 'climate forcing'. A gas isn't a 'force'. James_ wrote: None of these materials affect ozone the Chapman cycle. James_ wrote: These gases don't have that property. Ozone is created by UV light. It is destroyed by higher frequency UV light and it's own instability. As long as you have oxygen and sunlight, you WILL have ozone. You can't stop it. James_ wrote: Ozone is not being depleted. It never was. Man simply does not have the power to harm the ozone layer, even if we wanted to. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 29-09-2017 00:25 | |
| Into the Night (21582) |
Wake wrote:GreenMan wrote: James, I think you are right about this. And you point out something that I have been trying to get at. I recall Parrot Killer [not too good at it though, because Wake is still alive and doing well] saying something about the emissivity changing, but I wasn't quite sure what he was bitching about, because it wasn't me saying it. He was just twisting something around that I was saying, to make it look like that. But anyway, I don't think the emissivity has to change at all, to accommodate the Global Warming Theory. It stays the same because it is just a number that represents the proportionality between radiation and temperature. It does vary from object to object, due to the amount of radiant energy the object is transforming into thermal energy. Be calm, dude. He's trying to describe emissivity. You don't understand it either. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 31-07-2019 14:05 | |
| tmiddles (3979) |
Into the Night wrote:James_ wrote:It applies to all substances. Not just steel. OK so I'm actually asking Into the Night a question. Fair enough if you don't want to answer since I've been ignoring you so far. But you are the star of this topic. So here it is: Does Stefan-Boltzmann apply to solid, liquid and gas? |
| 31-07-2019 15:58 | |
| IBdaMann (14389) |
tmiddles wrote:Into the Night wrote:James_ wrote:It applies to all substances. Not just steel. The answer is "Yes." The Stefan-Boltzmann Law applies to all matter, always, everywhere in the universe. If it is matter then the Stefan-Boltzmann Law applies. I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 31-07-2019 18:39 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote:James_ wrote:It applies to all substances. Not just steel. All mass. It does not matter what state it's in. Solids, liquids, and gases all behave according to the Stefan-Boltzmann law. While a gas does not have what most people would call a radiating 'surface', it does nevertheless radiate. The surface is the gas molecules themselves. If you looked at a picture of the Earth in infrared, you will see a bright surface surrounded by a thin 'fog'. This is the atmosphere radiating infrared. Since the Earth is not one single temperature (and neither is it's atmosphere), the infrared that is radiated is over a fairly wide band. The Stefan-Boltzmann law states: radiance = Boltzmann constant * emissivity * temperature ^ 4 Where: * radiance is in watts per square meter of radiating surface, * the Boltzmann constant (a natural constant, essentially converting the equation to our units of measure), or 5.670373 * 10 ^ -8 when using meters and kelvins. * emissivity is a measured constant of how well the surface radiates, compared to a perfect radiator (an ideal black body, or one that has no ability to reflect light at all), and a perfect reflector (an ideal white body, or one that has no ability to absorb light at all), expressed as a percentage. 100% is the ideal black body. * temperature is in deg K. Emissivity is a very misunderstood value. It is actually a measured constant and not a variable. All real bodies are 'gray' bodies, some value between 0 (perfectly reflective) and 100% (perfectly black). The way you measure it is to first measure the temperature of the radiating body as accurately as possible, then measure the [i][radiated/i] light coming off of it (not any reflected light!), and comparing your readings to an ideal black body at the same temperature (the known value). Since ideal white bodies have no radiance we don't need to use them for comparison. This measurement must of course be carried out in complete darkness, and no radiance from harmonic emission is included. Blackbody radiance is not the only way to produce light. It is, however, the only way to produce light that is related to the temperature of an object. It is the way Earth cools by radiating into space. Conductive heating and convective heating are still important in terms of surface temperature, even though they can't heat space (yes, the space around Earth has mass in it. It's not empty). The atmosphere is mostly heated by the surface by conduction. Moisture in the ground and in the air immediately above it might condense out to liquid form and become a visible cloud if the air is cold enough and there is enough moisture. If so, you get what is called advection fog. It's why swamps and lowlands tend to develop fogs at night. To heat the atmosphere, the surface must cool. Energy is not free. Another way to heat the atmosphere from the surface is by radiance (i.e. the Stefan-Boltzann law). This is how CO2 in the air can also be heated. Again, the surface is cooled, since it takes energy to radiate in the first place. In all cases, the natural tendency is for the surface and the atmosphere to become the same temperature, then heat simply becomes zero. There is no further flow. Like the water level between two vessels that reach the same level because there is a tube between them allowing flow. Convection is also important. This is simply heating by moving the mass itself. All liquids (gases are considered liquids for this purpose), have convective heating. Warm air expands, and so contains less molecules than air around it. It is therefore lighter and begins to rise as a 'bubble' of hot air. The atmosphere cools as you gain altitude as well, and also loses pressure. The bubble of hot air can only rise so far, since it is rising into air of less pressure. This allows the molecules of our bubble to expand as well, and that means a cooler temperature. Temperature is the average kinetic energy of molecules. This is thermal energy. It is measured by what we call temperature. Heat is the movement of thermal energy, not the energy itself. Like a current in a river, it is the movement of the water, not the water itself. You cannot slow or trap heat because that is like slowing or trapping a current. A slow stream as a reduced current, not a 'slow' current. A fast stream has a high current value, not a 'fast' current. Current can be measured in gallons per hour. Similarly, heat can be measured in degrees per second. This is measured in watts or calories. Once everything is the same level, heat is zero. Once the level between two liquids are equalized, current is zero. Heat is the 'current' or flow of thermal energy, not the thermal energy itself. Real greenhouses reduce heat. Specifically, they reduce convective heat. They still allow radiant heat. They get warmer in the daylight because radiant heat is the primary way they get heated by the Sun, and the only way to cool off again at night. By morning, the greenhouse is the same temperature as the outside. Most greenhouses have heaters in them. Greenhouses are what the temperature of the Earth at that location would be if there was no convection and conduction such as what takes place in the open atmosphere. Wind is a result of convection. Inside the greenhouse, there is no wind. Storms are convection made visible. They are not caused by convection, they ARE convection (and a bit of moisture). There are no storms in greenhouses. A hurricane is nothing more than convection and a bit of moisture. All that destruction, flooding, wind damage, etc. is simply convection in action along with a bit of moisture. Storms help cool off the surface. I've presented a lot here, rather than just the Stefan-Boltzmann law itself. Much of what I've covered here is the 2nd law of thermodynamics as well. It is expressed as: e(t+1)>=e(t) where e is the entropy of any closed system (one that has consistently defined boundaries), and t is time (pick a unit). The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan Edited on 31-07-2019 19:30 |
| 31-07-2019 22:52 | |
| IBdaMann (14389) |
Into the Night wrote: All real bodies are 'gray' bodies, some value between 0 (perfectly reflective) and 100% (perfectly black). Just a minor wording quibble. In science, all real bodies are black bodies. There is no such thing as a "grey" body. All bodies are black bodies and the completely theoretical body of emissivity = 1.0 is an "ideal" black body. Obviously you get to express your ideas in any way you see fit, and you would not be the first to confuse the term "black body" (any body with thermal activity) to mean "ideal black body" and to thus conclude that emissivity must therefore refer to the color of the body, i.e. emissivity of 1.0 = "black" and emissivity of 0.0 = "white" therefore everything in between must be "gray" ... ... but it's not. All bodies with thermal activity are "black" bodies and the theoretical body of emissivity = 0 is "white" because it has no thermal activity. ... but again, it's just a wording quibble. The content of what you are expressing is fine. Into the Night wrote:Blackbody radiance is not the only way to produce light. It is, however, the only way to produce light that is related to the temperature of an object. Yes. Thermal radiation is the only light that is based on temperature, re: Planck's Law and Stefan-Boltzmann. Into the Night wrote: It is the way Earth cools by radiating into space. Yup. ... and the reason it can heat up from the sun, re: Kirchoff's law. You're hitting all the big ones. Into the Night wrote: Conductive heating and convective heating are still important in terms of surface temperature, even though they can't heat space (yes, the space around Earth has mass in it. It's not empty). If you are discussing the surface, i.e. the ground, the bottom of the atmosphere, etc... then it's really only conduction at play. The convection only affects the rest of the atmosphere above the surface. Sure, the convection causes cooler air to descend to the surface ... whereby conduction heats it at the surface. Into the Night wrote: Another way to heat the atmosphere from the surface is by radiance (i.e. the Stefan-Boltzann law). This is how CO2 in the air can also be heated. Again, the surface is cooled, since it takes energy to radiate in the first place. ... and you can mention at this point that once you know that the surface is heating the atmosphere, you can then dismiss all arguments of IR absorption by CO2 and "re-radiation downward to warm the surface" as egregiously violating the esteemed 2nd law of thermodynamics. Into the Night wrote: Convection is also important. This is simply heating by moving the mass itself. All liquids (gases are considered liquids for this purpose), have convective heating. I think you mean "fluids." All fluids experience convection because they are not bodies. Of course air is a fluid. Into the Night wrote: Temperature is the average kinetic energy of molecules. Nope. Temperature is the average amount of thermal energy per mass. Molecular kinetics is the result of temperature, it is not temperature itself. Yes, molecular motion is all we have to measure temperature, but that is because we do not have the ability to open up molecules and see how much thermal energy they each have. I'm not saying that we can't use the thermometers we've always used to measure temperature ... but it actually is an example of a proxy measurement. It happens to be a very good one ... but the point is that the motion is not the temperature itself. Imagine this: I have a stationary rock that is 28 degrees Celsius, through and through. I then sling-shot it toward a wall, increasing the average speed of all the molecules by 200 km/hour. How much has the rock's temperature increased? Answer: none. It has not acquired any additional thermal energy. Kinetic energy, yes ... thermal energy, no. I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 31-07-2019 23:48 | |
| tmiddles (3979) |
Into the Night wrote: And earlier: Into the Night wrote:GreenMan wrote: Greenman's confusion seems to dominate the discussions here. There is a preoccupation with the very surface of the dirt/rock/water of earth where our shoes touch and the gases begin. So we can agree that we are talking about an Earth that includes everything from the upper atmosphere down to the molten core. The area most of life is concerned with is the lower atmosphere just above the surface. Temperature is usually measured at 2M/6Ft above the ground as it's given for human applications. We are not concerned ONLY with the temperature of the surface, ground level of Earth, and it's just a small part of the system. Repeatedly, I know to your frustration, it's been claimed that the air heats the surface (something that could only happen briefly at sunset) which is really irrelevant to what we're discussing. Sound about right? |
| 01-08-2019 01:11 | |
| Into the Night (21582) |
IBdaMann wrote:Into the Night wrote: All real bodies are 'gray' bodies, some value between 0 (perfectly reflective) and 100% (perfectly black). Correct. I use the term 'gray' in a descriptive sense to get the idea across that not all bodies are ideally black. In science only the term 'black body' is used, and the number only indicates how 'black' it is. IBdaMann wrote: Again, I am using a 'layman's speech here, as opposed to the more technically accurate but somewhat confusing speech of science to the layman. IBdaMann wrote: The ideal 'white' body does not have to be absolute zero. The thermal activity is simply not determinable, since the body is a perfect reflector. In the end it makes no difference in setting the other end of the scale, and ideal 'white' bodies don't exist. You actually get into why this works later. IBdaMann wrote:Into the Night wrote:Blackbody radiance is not the only way to produce light. It is, however, the only way to produce light that is related to the temperature of an object. No. Descending air is heated as it descends because of the increasing pressure. This is the other side of convection. Given nothing else, it would equal the energy in the hot air bubble. While the risen air is up there, though, it's spread over a larger area (which itself is a lower temperature for the same mass), and can radiate into space over that larger area. In the case of the troposphere, it can also be affected by the cooling effects of the formation of ozone over that wider area. When it descends again, it heats up according to the ideal gas law just like always, but it lost energy while it was up there. It won't heat up to as high a temperature as before. In ocean convection, the same thing happens, but the surface forms the radiating (and conductive) interface to transfer energy. Water, being a liquid, doesn't behave like anything like an ideal gas, but it still expands when warm. IBdaMann wrote:Into the Night wrote: Another way to heat the atmosphere from the surface is by radiance (i.e. the Stefan-Boltzann law). This is how CO2 in the air can also be heated. Again, the surface is cooled, since it takes energy to radiate in the first place. That is correct of course. A colder gas cannot heat a warmer surface. IBdaMann wrote:Into the Night wrote: Convection is also important. This is simply heating by moving the mass itself. All liquids (gases are considered liquids for this purpose), have convective heating. Quite right. Thanks. I meant 'fluids'. IBdaMann wrote:Into the Night wrote: Temperature is the average kinetic energy of molecules. Quite right. Thank you for the correction. I was tending to use the 'bouncing molecule' model there. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 02:16 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: If you wear shoes! tmiddles wrote: Yes. We can also apply the same principles to any energy node of the Earth, such as the one you seem to be interested in. tmiddles wrote: The one you are interested in. The Stefan-Boltzmann still applies, and the laws of thermodynamics still apply. Remember, the surface temperature IS affected by what happens everywhere else on Earth as well, including the upper atmosphere and space near Earth. tmiddles wrote: Here is where the mistake is made. In the 2nd law of thermodynamics, all systems must be closed. That is, all systems must have a consistent boundary. Any energy source or sink from outside that system cannot be considered. If you consider ONLY the bit of air just above the surface as your system, you cannot consider anything that might heat it from outside (i.e. the Sun) or anything that might cool from outside (i.e. near Earth space). In other words, according to the 2nd law of thermodynamics, you cannot move the goalposts around. If you consider only surface temperatures, you cannot consider space, or how something might prevent thermal energy being lost to space. You also cannot consider how thermal energy might be gained from space (i.e. the Sun). Indeed, you cannot even consider the temperature of anything underground, such as Earth's core, volcanic activity, etc. The system MUST be closed. In other words, it MUST be consistent. Example: A refrigerator might appear to reduce entropy. It creates a colder environment in its box than in the midst of a warmer room. But it doesn't. That refrigerator can ONLY do what it does because energy is coming from a power plant to run the thing. In other words, the system INCLUDES the power plant as well. Entropy over the whole system is still increasing. There is no such thing as 'local' entropy. That is discussing a different system entirely, and has nothing to do with any other system. It is to make a false equivalence fallacy. Unplug that refrigerator, and you can legitimately no longer consider the power plant as part of the system. The inside of the refrigerator will reach the temperature of the room. Entropy still increases. The atmosphere near the surface is the same way. If you ONLY consider the surface temperature, you cannot consider anything outside that system that might affect that temperature. To do so is like considering the refrigerator as a 'local' entropy. It's just a false equivalence fallacy all over again, and a moving goalposts fallacy. In other words, considering ONLY the temperature of the air above the surface is to ignore everything else. Any temperature change you happen to notice cannot be considered. It just happens. It's a black box that you can no longer consider. This concept is also noticed by Kirchoff. It is why you can sum all the energy nodes of a system into one total. Kirchoff is simply stating that the sum total is made up of the parts of any system summed together. The system is therefore considered a whole unit. Again, that system is a closed one. It must be consistent. What Kirchoff has done is allow a way to relate a subsystem to be part of a larger system, but that larger system must be closed (consistent). Therefore, it really doesn't make sense to consider ONLY the air just above the surface of the Earth. There is a Sun. That is significant. There is the space around Earth. That is significant. There is the thermal energy within the Earth itself. That is significant. tmiddles wrote: It doesn't happen at sunset. The air cools off faster than the surface does. It has much less mass than the surface. tmiddles wrote: No. The only way air can heat the surface is if it is warmed somewhere else by an even hotter surface, and moves in over a colder surface. This can sometimes happen in a strong warm front, for example. Here in the Pacific Northwest, due to our proximity to the ocean, temperatures don't change much. Water has a high specific heat index. It takes more energy to warm 1 mole of water 1 degree than it does to warm 1 mole of dry air one degree. A flywheel takes a lot of energy to get started and spinning at a certain rate. A shaft without a flywheel is easier to get spinning at the same rate. The flywheel is not storing energy, the shaft it's mounted on will still lose energy, but it has a LOT more energy to lose, compared to the shaft without a flywheel. Water acts a bit like a thermal flywheel. It's why towns near the ocean tend to have a narrower temperature swing during the day than towns in the desert and not affected by all that water. An atmosphere acts a bit like a thermal flywheel in its own right. It is mass. It takes energy to heat it. It takes time to lose that energy. The thicker the atmosphere, the more like a flywheel it becomes. All mass also acts like a flywheel. On a planet with a very thin atmosphere, only the outer material is changing temperature so rapidly. Not very far down, temperature isn't changing much at all. You can use this fact to your advantage in the desert, and many animals do. The surface of the desert can get quite hot during the day, and quite cold at night. Most critters in the desert dig down just a foot or so and enjoy an almost constant temperature through the day and night. Life in the desert is best just a foot or two down. You can even make water in the desert that way. There IS humidity in the air, low as it is. If you have such a cave, you can suspend a tarp just outside the entrance, put a rock in the middle and a cup under the tarp where the rock is. Water will condense on the underside due to the temperature difference from your cave (and even sometimes in the open desert since the tarp is shade!), and drip into the cup. It's not much, but it can extend your water supply significantly if you are stuck out in the desert. Now we go back to Venus and Mercury, another interest of yours. Venus has a very thick atmosphere as you know, 90 times Earth pressure at the surface. This incredibly thick atmosphere formed from volcanic activity. It was the surface 'boiling off' gases that became the atmosphere of Venus. It was never like Earth, not even remotely. There is almost no hydrogen. No water, no life, no hydrocarbons, no nothing. The surface is hotter where we've measured it because it is darker. It stays hotter even at night because the atmosphere is such a good conveyor to carry thermal energy right around the planet. While the emissivity of Venus is unknown, it does seem to be higher than it's neighbor closer to the Sun, Mercury. In other words, Venus simply absorbs light better and converts that light to thermal energy. It is not as reflective as Mercury is. It takes time to heat the surface of Venus, but like that flywheel I described earlier, a LOT of energy is going into making Venus what it is. The thicker atmosphere only adds to the flywheel effect and better couples the warmer surface to the atmosphere above it. The day on Venus is long. A single day is like third of our year. That's a LONG hot day on Venus. It's also a LONG night, but the thick atmosphere helps to alleviate the effects of that by conveying daytime temperatures around to the night side. Mercury is closer, but it is more reflective as well. It also has no significant atmosphere to help move thermal energy around. While the surface has a thermal flywheel effect, those nicer temperatures are UNDER the surface, just like the deserts here on Earth, but more so. It is the same with our Moon. The top dirt is radically changing temperature. Down just a small amount makes an amazing difference. Note that CO2 simply has no effect here, other than being a mass. Be aware that this model is somewhat simplistic, but it gets the idea across. It ignores thermal energy by fission and internal pressures from tidal forces acting on the body in space. (We do know the core of the Moon is in the range of 2500 deg F. We don't know if it's due to fission or due to tidal stress.) It is also important to remember that flywheels do not store energy. It simply takes more energy to get them going, and they have more energy to give back as they slow down. The specific heat capacity of a substance is a good indicator of how much that substance will act like a 'thermal flywheel'. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan Edited on 01-08-2019 02:24 |
| 01-08-2019 04:31 | |
| tmiddles (3979) |
Into the Night wrote: OK all of that made complete sense and I'm following you. There are several interesting things there but plenty of time to get to them later. Let me present this to see if it jives with your understanding. For the sake of a clarity let's divide the matter which is EARTH into five layers: A- Upper atmosphere from 10 feet up to the void of space B- Lower atmosphere from the ground up to 10 feet C- The ground down one inch D- The ground from one inch down to one foot E- Below one foot. All five layers are one Earth, they cannot be considered independently, but I can present my understanding by talking about them as interacting parts of a whole. Also for the sake of a clear description let's consider only radiation/light from the sun, from infra-red to ultra-violet, impacting the ground as our only energy source. Let's ignore layer 5 and it's geothermal contribution as nothing more than a stabilizer for the system. Also let's only consider conduction/convection between the layers of EARTH and radiation off the EARTH entirely. Let's ignore radiation between the layers. So here is the journey of energy: 1. Energy which already existed is release from the sun in a fusion reaction sending radiation from infra-red to ultra violet to earth (entropy increases) 2. The radiation strikes the surface, layer C, converting to thermal energy in vibrating the molecules of dirt. As the surface has more thermal energy than the layer of air above or the layer of dirt below, "heat" the movement of thermal energy from hotter to colder, results in conduction of thermal energy and it's movement from the surface down into the dirt as well as up into the air. 3a. Layer D, just below the surface has a rising temperature as thermal energy moves down into it from the always hotter surface layer C. 3b. Layer B, the air just above the surface, has thermal energy move up into it as gas molecules make contact with the hotter surface. 4. Convection of warm air allows the warm air from layer B near the ground to move relatively quickly up into the upper atmosphere layer A. While the temperature of an individual molecule of gas increase (moves more) what we call the temperature (the amount of thermal energy within a unit of space?) drops as the pressure/density of gas drops. 5. All of the matter, but primarily the surface layer C, and the upper atmosphere layer A, release the the thermal energy back out into space in the form of infra-red radiation. (entropy increasing the whole time) At night the input of energy is gone, and the surface of the earth cools. Thermal energy from layer D, which originally moved downward from the surface, now moves up into the surface layer C because the lower atmosphere is now so much colder. However the surface layer C still has more thermal energy and is warmer. All of the thermal energy that enters the system Earth in 24 hours has a corresponding and equal amount of energy leaving Earth as infra-red radiation (if you looked at the Earths dark side with infra red goggles you'd see it glowing). There is thermal energy that's present, we don't go down to 0 at night, because of the flywheel way in which the matter has a "lag time" in the thermal energy passing through it. For a very stripped down model sound correct? Edited on 01-08-2019 04:32 |
| 01-08-2019 04:40 | |
| IBdaMann (14389) |
Into the Night wrote:The ideal 'white' body does not have to be absolute zero. Yes it absolutely must be absolute zero. It cannot absorb any thermal radiation and therefore has zero thermal energy. Per Kirchoff's law, even if it were to somehow have thermal energy it would not be able to emit any of it. A white body is incapable of thermal activity. It has no thermal energy. It is absolute zero. Into the Night wrote: I'm sorry but the surface is solid and does not experience convection. All convection is in the rising air. Rising air heats other air via convection and the surface can only heat air via conduction. I suppose you could say that the surface increases atmospheric temperature indirectly via convection, with the rising air being the direct cause of convective heating. Sure, I'll buy that. Into the Night wrote:Quite right. Thank you for the correction. I was tending to use the 'bouncing molecule' model there. The interesting thing about a molecule having more thermal energy from having absorbed some photons is what that actually means. We had a discussion previously about how people tend to envision atoms as little balls flying around, bouncing off each other in elastic collisions, and when the temperature increases, they bounce around "faster." The reality is that each atom/molecule is a fluctuating probability field, with higher probabilities clustered more densely around a particular point with probabilities decreasing as you extend outward from that point. As the temperature increases, the probability values themselves expand outward, explaining how matter expands with increased temperature, highlighting how different materials expand differently because their probability spaces change differently with increases in temperature. When the volume of a gas is reduced, the probabilities become more evenly distributed throughout the probability space, representing an increase in pressure because of increased probability overlap with other atoms/molecules. So when two atoms collide, what you have is not two balls bouncing off each other but rather two probability fields affecting each other in ways that are not necessarily straightforward. If you think about it for a while you will yearn for the simple "bouncing ball" model ...  I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 01-08-2019 04:51 | |
| tmiddles (3979) |
IBdaMann wrote: As the temperature increases, the probability values themselves expand outward, explaining how matter expands with increased temperature, So my understanding is that the individual molecules of gas have more thermal energy higher in the atmosphere than lower in it. Because of this they expand, take up more space and are less dense, so there is less thermal energy per volume of space higher up even though the gas is hotter? |
| 01-08-2019 05:08 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: Up to a point. tmiddles wrote: I assume you mean layer E. Fine. We can ignore that layer. We can simply declare it not part of our system, which consists of layers A-D. tmiddles wrote: You can't. It is part of layers A-D. For this purpose, I'll let you summarize around it for now though. tmiddles wrote: Oops. You've just added a layer F, which includes the Sun. You haven't gotten very far, though, so fine so far. tmiddles wrote: Since you specifically mentioned 'ground' as layer C and ignored the oceans, fine. tmiddles wrote: Fine. tmiddles wrote: Fine. tmiddles wrote: Per mass, actually. IBdaMann is correct about that one. However, yes. Gases cool as they expand, according to the ideal gas law. tmiddles wrote: Actually primarily surface layer C. The upper atmosphere is quite thin. It radiates, but it is not most of the radiance, not by a long shot. For the purposes of this discussion, that is the only correction here. Otherwise, you are correct. tmiddles wrote: Fine. tmiddles wrote: You will see the same thing in the daytime as well. Earth is always radiating. Day and night. tmiddles wrote: Not a lag time. Flywheels don't store energy. They just have more energy to give up. Same with the surface for this discussion. tmiddles wrote: Much closer. You are making great progress to understanding this. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 05:29 | |
| Into the Night (21582) |
IBdaMann wrote:Into the Night wrote:The ideal 'white' body does not have to be absolute zero. No. Nothing says it has to absorb light to gain its thermal energy. To say an ideal white body has no thermal energy at all is to say there is no body at all, not even a theoretical one. Mass, energy, and light all become void at absolute zero, for there is no speed of light there that means anything. Since E=mc^2, the equation reduces to 0=0*(the speed of light?) squared. Energy is zero. Mass is zero. The speed of light in a vacuum is a speed, relative to something. That is energy, but energy is zero. So is any reference point, such as a mass. Absolute zero is an ideal that can never be achieved, that we can detect anyway! IBdaMann wrote:Into the Night wrote: I hear ya there! Fortunately, we can still use the old models for describing certain things in theories that used these older models as part of their formation. At least, despite it all, the theories themselves have not yet been falsified, and they were formed around older models. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 05:29 | |
| IBdaMann (14389) |
tmiddles wrote:So my understanding is that the individual molecules of gas have more thermal energy higher in the atmosphere than lower in it. No. A molecule's quantity of thermal energy is strictly a function of how much electromagnetic radiation it has successfully absorbed. Altitude is not a factor. Molecules at the top of earth's nighttime atmosphere are of the same temperature as the moon's nighttime surface. Molecules at the top of the earth's daytime atmosphere are of the same temperature as the moon's daytime surface. Altitude will affect the air pressure ... which will affect the amount of mass per volume. So yes, at higher altitudes there is less thermal energy per given volume even though you might have individual molecules/atoms of higher amounts of thermal energy (temperature). Look at it this way, your body "feels" hot and cold by sensing thermal energy flowing in/out of your body (actually your skin is very good at this). If you were to somehow fly up to the very top of earth's daytime atmosphere where the individual air molecules are as hot as the moon's daytime surface, they would nonetheless not feel hot at all because 1) there is zero atmospheric pressure, 2) there are so few molecules in which you can come into contact at any given moment and 3) the sum total of thermal energy flowing into your body is negligible and in no way enough for your body to sense it. Ergo there is negligible thermal energy yet the individual molecules are of very high temperature. This makes [part of] your next assertion correct: tmiddles wrote: [incorrect part deleted]... are less dense, so there is less thermal energy per volume of space higher up even though the gas is hotter? Absolutely. I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 01-08-2019 05:32 | |
| Into the Night (21582) |
tmiddles wrote:IBdaMann wrote: As the temperature increases, the probability values themselves expand outward, explaining how matter expands with increased temperature, No. They do not gain thermal energy by expanding. They are simply expanding because of the thermal energy. There is less mass per volume, which means that parcel of air lighter and begins to rise, just like a hot air balloon. At altitude, there is less thermal energy per volume. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan Edited on 01-08-2019 05:35 |
| 01-08-2019 05:33 | |
| tmiddles (3979) |
Into the Night wrote: So yes it makes sense that the surface would radiate more as it has the most thermal energy in it and it has more matter. Though if the entire atmosphere was condensed into a liquid it would be 10 meters deep so it could be very comparable in radiating as there is a lot of it all with a clear shot to send radiation into space. The radiation passes right through almost all of the air so the air doesn't get in it's own way where the dirt does. I guess it's a question of how much conduction with air, a poor heat conductor, can occur at the surface. Now how can the "Flywheel", hang time, storage, be properly described. Would it be fair to say that in the system of the earth, if you take a first in last out perspective on thermal energy, that the flywheel effect of the ocean and the system as a whole is basically the thermal energy currently within the system, the thermal energy "in residence" which though it's on it's way out hasn't left yet. In my example above it started at the surface and moved through both dirt and gas before finally radiating back out into space. A stasis/equilibrium exists so this amount just fluctuates within a fairly narrow range. The same stabilization a flywheel provides in a mechanism. I understand all of this much more after being on the forum a bit and having to look everything up. We are finding common points of understand to make a discussion possible which is great. Edited on 01-08-2019 05:51 |
| 01-08-2019 05:50 | |
| tmiddles (3979) |
IBdaMann wrote:there is negligible thermal energy yet the individual molecules are of very high temperature. Into the Night wrote: Got it. Basically I figured if they did "cool" they would sink back down into the atmosphere to be replaced at the top by "hotter" gases. I'm just seeing there is a bit of disconnect between the everyday notion of "temperature", it's hot or cold to my hand, and the thermal energy in a molecule. A lot of thermal energy could be moving through some very "hot" molecule at the top of the atmosphere but a thermometer would tell you it's freezing. |
| 01-08-2019 05:51 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: Thermal energy is always moving. There is always heat. This is true even through thermal insulation, like blankets or sweaters. There is just less heat (less flow). No insulator is perfect. Flywheels do not store energy. They simply have more energy for a given shaft speed than a shaft without a flywheel. It takes more energy to get the thing turning, and more energy is available as it slows down again. We are basically talking inertia here in our comparison. Like throwing a pebble vs a heavy rock the same distance. One simply takes more energy to do it. That same one makes more of a thud when it hits the ground again. Anything with thermal energy has a similar kind of 'inertia' (let this one go, IBdaMann, I know I am making a simplistic model here). That thermal energy will tend to stay the same, unless it has somewhere to flow to. It is that flow we call 'heat'. Since there is no perfect insulator, there is always heat. Specific heat capacity indicates this 'inertia'. Water, for example, has a high specific heat capacity. Water one degree warmer got there by absorbing more thermal energy than dry air did. It gives up that same thermal energy when it cools by one degree, more than dry air does. But it's always warming or cooling. There is always something warming or cooling it. There is always heat. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 05:56 | |
| tmiddles (3979) |
Into the Night wrote: Since I know you object to calling the energy that has "built up" in this way "stored" what would you say? What's a good way of saying that the thermal energy is present in some quantity? The temperature at night or in the shade would be from this quantity of accumulated thermal energy. If we are talking about temperature it seems it comes down to the input of energy from the sun (or the degree to which the suns energy has the ability to be transmitted to a particular place at a particular time) and this latent/accumulated thermal energy moving though the system. Edited on 01-08-2019 06:15 |
| 01-08-2019 08:50 | |
| Into the Night (21582) |
tmiddles wrote:IBdaMann wrote:there is negligible thermal energy yet the individual molecules are of very high temperature.Into the Night wrote: It will cool to the temperature of the rest of the air up there. Only if a cold bubble of air forms in warmer air around it will it descend on its own. tmiddles wrote: Actually the thermometer will tell you the correct temperature. You, however, will think it's freezing. This has to do again with the fact that you are warm blooded. Your body maintains it's own temperature using the food you eat. At the upper atmosphere, there is very little pressure, and really nothing for you to feel with thermal energy, compared to your own body temperature. Thermometers don't have a body temperature to maintain, so it will show the actual temperature there, not what it feels like to you. Yeah, I know it's weird. Of course, the cold feeling air is the least of your worries. You will pass out and die for lack of oxygen before you feel much anything else. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 08:52 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: Temperature. tmiddles wrote: It IS the temperature. tmiddles wrote: The temperature shows how much thermal energy you have. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 08:57 | |
| tmiddles (3979) |
Into the Night wrote: You're talking about wind chill right? 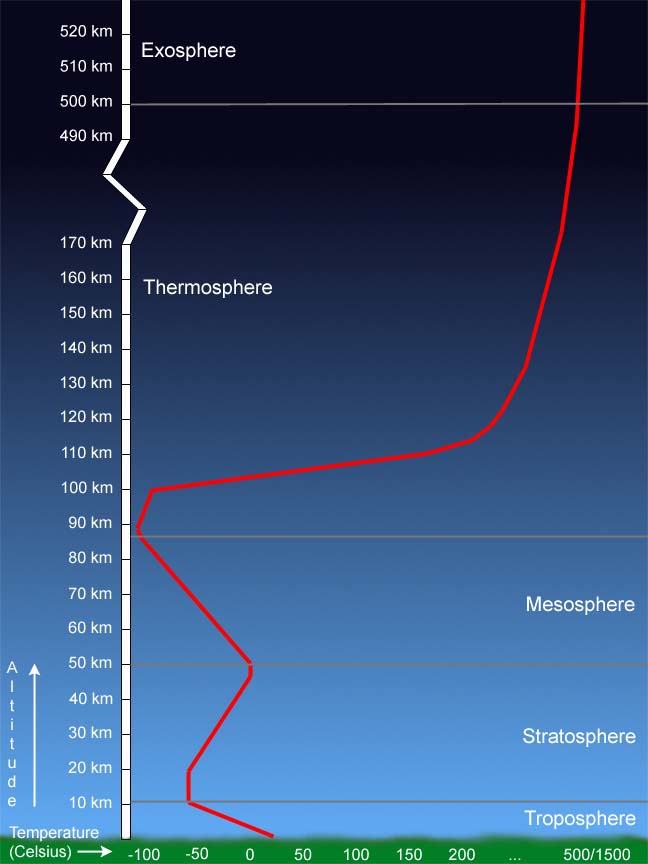 So I just looked I had no clue. I didn't know it got so hot up really high. I just knew about the cold part (someone freezing hiding in jet plane landing gear) |
| 01-08-2019 09:00 | |
| tmiddles (3979) |
Into the Night wrote: A piece o tin foil and a 100lb slab of steel can both be 200C but the slab of steel has more thermal energy. How would you describe that? The flywheel aspect of available thermal energy Like the difference between specific heat and just heat Like a Btu is the amount of heat necessary to raise 1 lb. of water through 1 degree Fahrenheit. A calorie is the amount of thermal energy needed to raise the temperature of 1 gram of water by 1 degree Celsius. That's not the same as giving the temperature in degrees of something. Edited on 01-08-2019 09:09 |
| 01-08-2019 09:25 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: No, I am not talking about wind chill. Like wind chill though, it feels colder than it actually is. Airlines generally fly at altitudes from 30,000 ft (FL300) to 45000 ft (FL450). On your chart that's around 9 to 14 km. You will notice that is the location of the tropopause, one of the coldest parts of our atmosphere. You will notice it's often around -50 deg C (about -58 deg F) at that altitude. Now you know why it's so dangerous to stow away in landing gear. There's not enough oxygen so you pass out in about 30 seconds up there and then freeze to death. Usually the body falls on whatever is underneath the aircraft when it lowers its gear at the outer marker of the approach (it falls 2000 ft when the gear is lowered). Being frozen, it just sort of bounces a bit and scares the crap out of whoever witnesses the thing coming out of the sky. If things are lucky, the body remains stuck to the gear strut, and the mechanics find it when they service the plane. They chip it off the strut and call the police. Then they clean up the strut and return the plane to service. Certified aircraft mechanics have some really gross jobs sometimes. Airlines fly there because of three reasons: 1) The weather rarely gets that high. They can fly above it most of the flight. 2) The air is thin, reducing drag. 3) The air is cold, allowing the jet engine to compress it more efficiently and create a greater difference between the hot and cold sections of the engine (making it more efficient). Flying at altitude, the jet engine is the 2nd most efficient engine we've ever made. The 1st is the diesel electric locomotive. A single locomotive can move a whale of a lot of freight. The only reason the jet engine isn't in 1st place is because it's a real fuel hog getting up there in the first place and when taxiing on the ground. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan Edited on 01-08-2019 09:38 |
| 01-08-2019 09:41 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: The slab of steel has more mass than the tin foil. There is more mass to heat. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 09:48 | |
| tmiddles (3979) |
Into the Night wrote: Well I don't mind "stored thermal energy" or "pent up thermal energy" but how would you like to describe the amount of thermal energy, originally from solar radiation, that is in the Earth system? Temperature doesn't work because it also just means the thermal energy at a particular point. Tin foil and the slab of steel can have the same temperature. We could just say the BTUs of thermal energy. Or how about: "The calories of earth" : ) Edited on 01-08-2019 09:49 |
| 01-08-2019 10:02 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: There is more thermal energy in the slab of steel because it has more mass. Remember the temperature also includes the mass you are taking the temperature of. No mass, no temperature. A 100 lb chunk of tinfoil and a 100lb chunk of iron, pretty much the same thermal energy at the same temperature. It's like getting hit by a BB or getting hit by a bowling ball moving at the same speed. The BB might sting, but the bowling ball will kill you; yet they both have the same speed. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 01-08-2019 10:20 | |
| tmiddles (3979) |
Into the Night wrote: Two objects can have the same temperature and different quantities of thermal energy? Temperature describes the intensity not the amount. It's like voltage doesn't describe the amount of electrical energy. You need to know the Amps and then watts. So what language works for you? "As a form of energy, heat has the unit joule (J) in the International System of Units (SI). However, in many applied fields in engineering the British thermal unit (BTU) and the calorie are often used. The standard unit for the rate of heat transferred is the watt (W), defined as one joule per second." Thermal potential energy? Edited on 01-08-2019 10:26 |
| 01-08-2019 21:05 | |
| IBdaMann (14389) |
tmiddles wrote:A lot of thermal energy could be moving through some very "hot" molecule at the top of the atmosphere but a thermometer would tell you it's freezing. Correct. A thermometer is an example of a proxy measure, i.e. it measures something other than what we claim it is measuring. Temperature is determined by quantity of thermal energy but thermometers measure the aggregate kinetic energy of the molecules striking it, i.e. something completely different, and then we turn around an claim that we just measured thermal energy, ... which we didn't. Kinetic Energy is not Thermal Energy. So if extremely few, but very hot, molecules strike the bulb of a thermometer, they will only be able to generate extremely little kinetic energy in the mercury which will then cause the thermometer to indicate an extremely cold temperature. But wait! Weren't the molecules extremely hot? Yes, of course. The thermometer is simply measuring the kinetic energy and not measuring the thermal energy. It should be called a "kinetometer" but it's not. So, let's put on our scientist caps and apply the scientific method to the existing science. Maybe they got it wrong! If I have a kinetometer, er, I mean a thermometer in a sealed container filled with a gas at pressure P, and I am getting a reading of temperature T, ... and then I simply decrease the pressure by half but not changing the temperature of the molecules, the Ideal Gas Law predicts that the temperature will decrease. But wait, didn't the temperature of the molecules remain the same? Why does the science predict a temperature decreases when the molecules' temperature obviously remains the same? Answer: Because the assumption is that the temperature is being measured by a kinetometer and the law predicts a decrease in the total kinetic energy which is being equated with thermal energy ... because that is the only way we can measure temperature ... by proxy. I don't think i can [define it]. I just kind of get a feel for the phrase. - keepit A Spaghetti strainer with the faucet running, retains water- tmiddles Clouds don't trap heat. Clouds block cold. - Spongy Iris Printing dollars to pay debt doesn't increase the number of dollars. - keepit If Venus were a black body it would have a much much lower temperature than what we found there.- tmiddles Ah the "Valid Data" myth of ITN/IBD. - tmiddles Ceist - I couldn't agree with you more. But when money and religion are involved, and there are people who value them above all else, then the lies begin. - trafn You are completely misunderstanding their use of the word "accumulation"! - Climate Scientist. The Stefan-Boltzman equation doesn't come up with the correct temperature if greenhouse gases are not considered - Hank :*sigh* Not the "raw data" crap. - Leafsdude IB STILL hasn't explained what Planck's Law means. Just more hand waving that it applies to everything and more asserting that the greenhouse effect 'violates' it.- Ceist |
| 01-08-2019 22:24 | |
| Into the Night (21582) |
tmiddles wrote:Into the Night wrote: Yes. Temperature is the intensify within a given mass. In other words, larger masses have more thermal energy for the same temperature. tmiddles wrote: It describes the force. Electrostatic force. It is like pressure in a pipe. tmiddles wrote: Amperage is current. It is like heat of the current in a river. It is not the charge itself. One ampere is one coulomb of charge going past a point in a circuit per second. A coulomb is a specific amount of energy (charge). Watts is a measure of work, or energy. One watt is one joule per second, or one coulomb of charge being forced to move through a point in a circuit by the force of one volt. A joule is the capacity to do work. It is not the force, it is not the movement. It is the combination of the two. One joule is the capacity to move 1 newton 1 meter in the direction the force is applied. A watt is doing just exactly that in one second. The capacity to do it is energy. Actually doing it is expending that energy as work. tmiddles wrote: Heat is not measured in joules. It is the flow of energy, not a force. It is not energy. Heat is measured in calories, or in watts. Thermal energy is energy. It is the capacity to do work. It is measured in joules. It is the average kinetic energy of the particles within a substance, not the kinetic energy of the substance as a whole. Temperature is the average kinetic energy of the particles within a substance. If there is more substance, there are more particles, and the average can stay the same. But since there are more particles, thermal energy is greater for the same temperature. tmiddles wrote: Nope. Just energy. It is the capacity to do work. Kinetic energy and potential energy are both energy. They both have the capacity to do work. Temperature is a useful indicator of thermal energy when the same mass is used. The Parrot Killer Debunked in my sig. - tmiddles Google keeps track of paranoid talk and i'm not on their list. I've been evaluated and certified. - keepit nuclear powered ships do not require nuclear fuel. - Swan While it is true that fossils do not burn it is also true that fossil fuels burn very well - Swan |
| 02-08-2019 00:10 | |
| tmiddles (3979) |
IBdaMann wrote:So if extremely few, but very hot, molecules strike the bulb of a thermometer, they will only be able to generate extremely little kinetic energy in the mercury ...It should be called a "kinetometer" but it's not. Or even "contact kinetometer" since the inability to measure even a small volume is a big issue too. You are using the information from a thermometer, assuming you know the consistency of what you're measuring, to then calculate the kinetic energy? So we are still missing a descriptor here: The information from a thermometer tells us how much kinetic energy has molecules crashing into the sensor at one location in space. We are forced to make assumptions about that the kinetic energies consistency in the volume being evaluated. We then have to use the pressure to calculate the kinetic energy per mole of gas. This could show that per molecule the energy is actually higher at a lower pressure than at a higher pressure where the thermometer show a higher reading. This is why saying two gas volumes have the same temperature wouldn't be useful as they could have very different amounts of kinetic energy. We are missing a good way to describe the quantity of the thermal energy. The sum the potential of all that vibration in the molecules. It would not be a measurement relative to time like a Watt because it's not going anywhere necessarily. It's not "Heat" because that's the movement of IT not IT. I think something like BTU is probably closest. Into the Night wrote:One ampere is one coulomb of charge going past a point in a circuit per second. A coulomb is a specific amount of energy (charge). So does joule work as the best unit for "Thermal Energy" In discussing the Earth system and the ground you can say that the temperature of the ground changes and that's useful since the mass doesn't change. But if you talk about the energy in the ground and the energy in the first meter of air above it and so on you then start to need a basis for comparison. Or different patches of ground. Edited on 02-08-2019 00:10 |
Join the debate Greenhouse Gases Do NOT Violate The Stefan-Boltzmann Law:
Related content
| Threads | Replies | Last post |
| The "radiative Greenhouse effect" does not exist | 140 | 15-04-2024 19:43 |
| 'Greenhouse' Effect? | 49 | 30-11-2023 06:45 |
| The SCIENCE of the "Greenhouse Effect" | 291 | 05-11-2023 22:46 |
| 17 year old cyclist murdered, do not expect the law to investigate, as the cyclist is always at fault | 0 | 31-07-2023 22:23 |
| Nitrate Reduction - Powerful Greenhouse Gas Emission AND Alkalinity | 102 | 05-06-2023 13:19 |